Let’s look at the water fluoridation issue.
The American Dental Association is still promoting water fluoridation to reduce the incidence of tooth decay. Proponents of water fluoridation claim that anti-fluoridationists are “fear mongers” who don’t understand the science.
Who is right?
Putting fluoride in the drinking water is a good idea if:
1. Ingesting fluoride internally in the water supply reduces cavities,
2. AND there was not an alternative way to deliver fluoride to the teeth,
3. AND if decay rates were significantly lower in water fluoridated communities than non-fluoridated communities,
4. AND there were no negative health consequences to ingesting fluoride,
5. AND you could control the dose,
6. AND people had the option of refusing fluoride in their drinking water.
It does seem that people living in communities with fluoridated water have shown a decrease in decay rate. Sounds pretty good. But wait, it turns out that a very similar decline in tooth decay incidence has also occurred in communities that do not fluoridate their water. See the chart below:
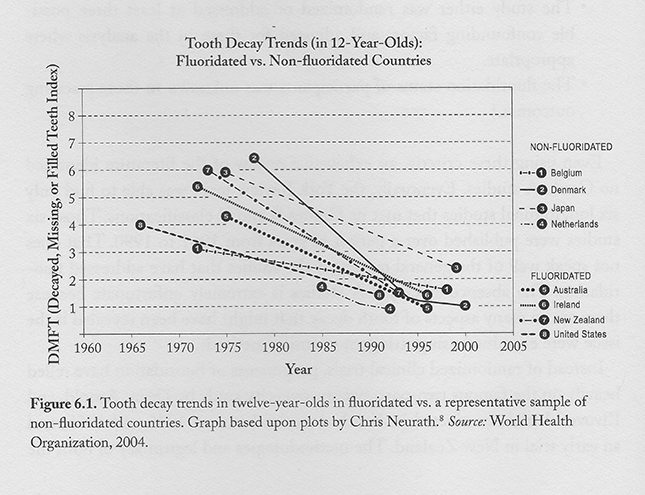
Paul Connet, James Beck, H.S. Micklem, “The Case Against Fluoride,” 2010, ISBN 978 1-760358-287-2, Chelsea Green Publishing.
Proponents of water fluoridation state that when fluoride is ingested, it gets incorporated into the tooth structure of developing teeth making teeth more decay resistant. But that does not seem to be the case. Except for first molar pit and fissure decay that can easily be prevented with the application of dental sealants, the decay preventive mechanism of fluoride action is entirely topical.
From www.cdc.gov.
…In the earliest days of fluoride research, investigators hypothesized that fluoride affects enamel and inhibits dental caries only when incorporated into developing dental enamel (i.e., preeruptively, before the tooth erupts into the mouth) (30,31). Evidence supports this hypothesis (32–34), but distinguishing a true preeruptive effect after teeth erupt into a mouth where topical fluoride exposure occurs regularly is difficult. However, a high fluoride concentration in sound enamel cannot alone explain the marked reduction in dental caries that fluoride produces (35,36). The prevalence of dental caries in a population is not inversely related to the concentration of fluoride in enamel (37), and a higher concentration of enamel fluoride is not necessarily more efficacious in preventing dental caries (38).
The laboratory and epidemiologic research that has led to the better understanding of how fluoride prevents dental caries indicates that fluoride’s predominant effect is posteruptive and topical and that the effect depends on fluoride being in the right amount in the right place at the right time. Fluoride works primarily after teeth have erupted, especially when small amounts are maintained constantly in the mouth, specifically in dental plaque and saliva (37). Thus, adults also benefit from fluoride, rather than only children, as was previously assumed.
It is well established that fluoride’s mechanism of action in reducing tooth decay is topical and not systemic.
Water fluoridation proponents go on to say that it is important to ingest the fluoride because once ingested, the fluoride ion will be slowly released in the saliva providing a constant source of topical fluoride. Well, that is not correct either. It is true that fluoride is found in saliva after drinking fluoridated water but the concentration is too small to have any effect at all on decay prevention. Studies show a saliva concentration of 0.016 mg./L fluoride in people who drink 1ppm fluoridated water. This concentration is significantly below the 0.04 mg/L fluoride that has been shown to be necessary to have any positive effect on decay prevention. Below 0.02mg/L fluoride there is no protective effect. So the claim that fluoridated water continually prevents tooth decay by providing a constant source of an effective concentration of fluoride in saliva is false.
Which leads us to our next point. Is there another way to deliver fluoride topically other than drinking fluoridated water?
Of course there is. Just brush your teeth with fluoridated toothpaste. If you brush your teeth regularly with fluoridated tooth-paste there will be a reservoir of fluoride in the mouth that will deliver the decay preventive effects of fluoride without having to ingest it.
The clearance of fluoride from saliva following toothpaste use has been investigated in a number of studies. Duckworth et al. [1991] found that following a single brushing the salivary fluoride level decreased in two distinct phases. There was an initial rapid phase (40-80 mins) followed by a slow phase that lasted for several hours and was possibly due to release of fluoride from intra-oral reservoirs. Repeated regular brushing led to elevated baseline values and the fluoride stored in the oral reservoirs was postulated to maintain a prolonged caries-protective effect. (emphasis ours)
We have already established that the effect of fluoride on tooth decay reduction is topical and not systemic and that the decay rate in fluoridated and non-fluoridated communities are essentially the same.
We conclude that the widespread use of fluoridated tooth- paste delivers an adequate amount of topical fluoride to cause a reduction in tooth decay. There is no need to deliver fluoride in the water. The argument that fluoridated water provides a topical source of fluoride may be true, but it is not superior to topical fluoride application achieved through regular tooth brushing.
There are many red flags regarding the negative health effects of fluoride.
One outward sign of excess fluoride intake is dental fluorosis. Dental fluorosis, also called mottling of tooth enamel, is a developmental disturbance of dental enamel caused by excessive exposure to high concentrations of fluoride during tooth development. The CDC reports that 41% of children in communities where the water is fluoridated have dental fluorosis. Dental fluorosis is a visible effect of too much fluoride. If the fluoride is damaging the tooth enamel what is happening to internal structure that we cannot see like the brain, thyroid gland and bones?
With fluoridated water you cannot control the dose.
A liter of water contains 1 mg of fluoride. Some people drink 1 liter of water per day while others can drink 2,3 or 4 liters of water /day. Why would we want to mass medicate a population of people and not control the dose?
A glass of water contains 0.25 mg of fluoride. A pea size amount of fluoridated toothpaste also contains the same 0.25 mg. of fluoride. On the back of toothpaste tubes there is a warning that says if this pea sized amount of toothpaste is swallowed, contact poison control or your physician. This pea size amount of toothpaste contains the same amount of fluoride that is found in a glass of water form a fluoridated water source that is fluoridated with 1ppm fluoride.
There are many red flags regarding the safety of fluoride. Fluoride is NOT an essential nutrient. Concerns about cognitive effects such as reduced IQ, bone cancer, thyroid dysfunction, skeletal abnormalities are just some of the systemic health concerns regarding fluoride consumption.
Further, you can’t control the dose. Some people drink a lot more water than others. So people that drink a lot of water will be consuming much more fluoride than somebody who drinks much less water. Also, how can you mass medicate a population when you can’t control the dose? And should we be mass medicating people at all?
What about people who are extra sensitive to fluoride like the elderly with kidney disease? Why should they be subjected to fluoridated water?
Putting fluoride into the public water supply simply makes no sense
Because:
- You cannot control the dose.
- Fluoride works topically and not systemically.
- You can get the same decay protective effects of fluoride by regularly brushing with fluoridated tooth-paste. The effect and delivery will be strictly topical.
- Decay rates have been decreasing at just about the same rate in both fluoridated and non-fluoridated communities.
- There are many red flags on the negative health effects of fluoride ingestion.
To investigate both sides of this topic go to:
The following video is a debate between dentist Dr. Howard Farran and Dr. Paul Connett, Professor Emeritus of Chemistry at St. Lawrence University.
An Inconvenient Tooth- Fluoride Documentary.
Bibliography: